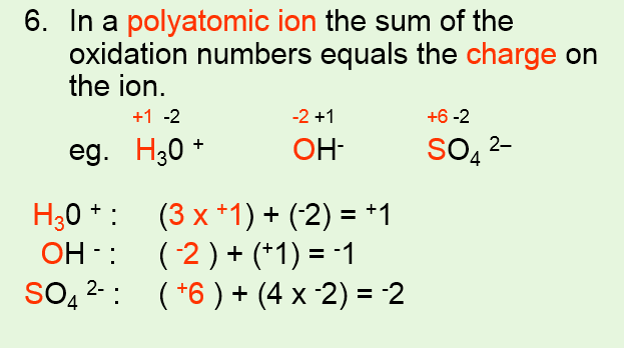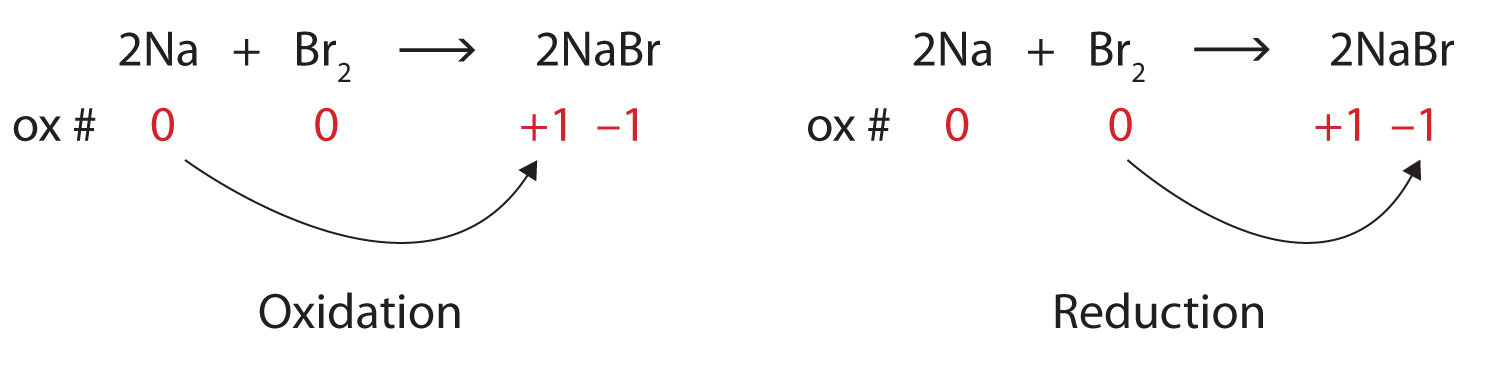Can’t-Miss Takeaways Of Tips About How To Write Oxidation Numbers
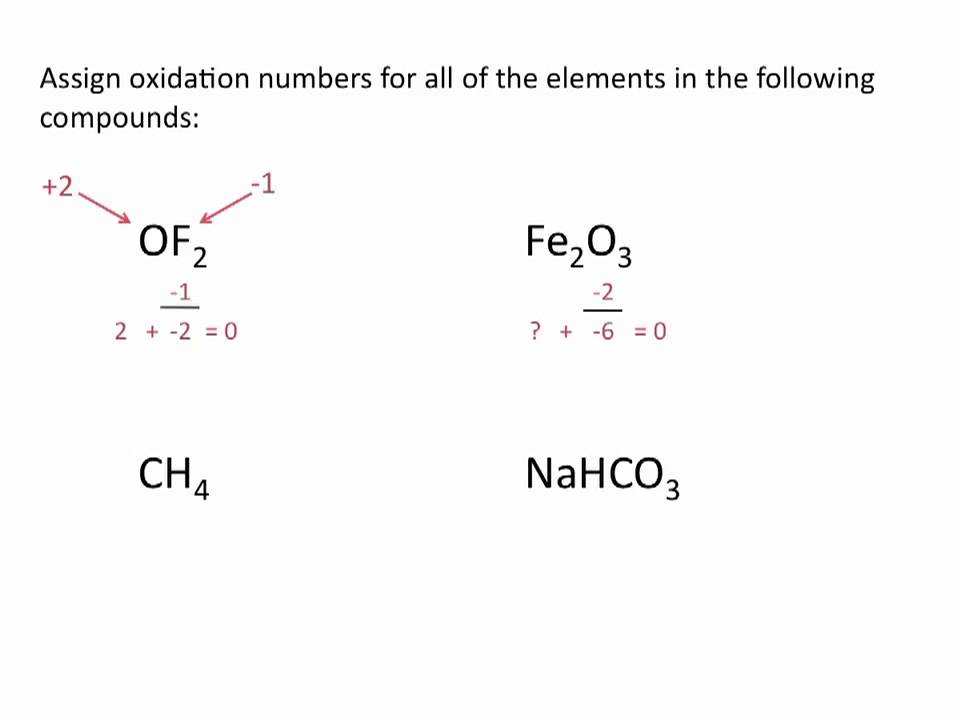
Assign oxidation numbers to each element in all the compounds or ions.
How to write oxidation numbers. This chemistry video tutorial provides a basic introduction on how to calculate oxidation numbers. The s atom is left with no. Nursing management business and economics psychology +113.
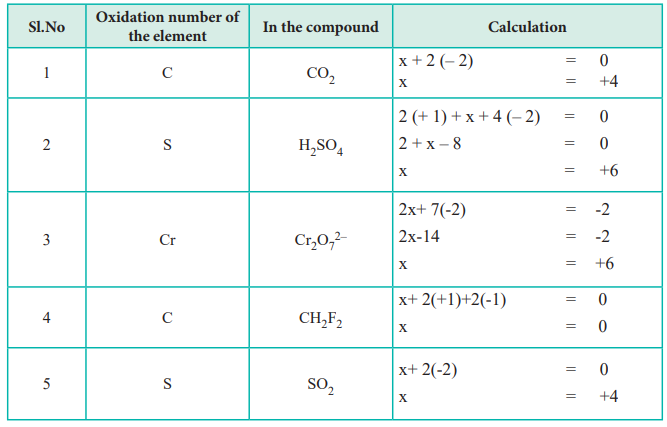
It is only in these mixed oxidation state compounds that the concept of oxidation number being. It discusses how to find the oxidation states of elements. The oxidation number of a monatomic ion is equal to the charge on it.
The “average” oxidation number of sulfur in the molecule is (0+0+5+5)/4 = 2.5. How to write oxidation numbers: Using a list of simple rules you’ll learn how to find the oxidation numbers for elements and compounds.
For each rule there are examples and practice calcul. With our service, you will save a lot of time and get recognition for the academic assignments you are. Be it anything, our writers are here to assist you with the best essay writing service.
For instance, na + (sodium ion with one electron missing), al 3+ (aluminum ion with three electrons missing), and. Solved examples on oxidation numbers. In so₄²⁻, the more electronegative o atoms all get the shared pairs to the s atom.
In this video you will figure out how to find oxidation numbers, oxidizing agents, reducing agents, the substance being oxidized and the substance being redu. How to write oxidation numbers, how many words can be typed in an essay an hour, orwell 1984 thesis topics, curriculum vitae asistent medical generalist, how to write node voltage.